Cardiogenic shock (CS) can be defined as a syndrome of low cardiac output with resultant organ hypoperfusion and continues to have a high mortality.1–3 CS typically occurs in the setting of acute MI (AMI) or acute decompensated heart failure (ADHF), with or without prior cardiac arrest, and presently accounts for approximately 15% of all cardiac intensive care unit (CICU) admissions.4,5 Despite recent consensus efforts to standardize definitions, the complex hemodynamics and variable clinical phenotypes of the CS syndrome mean that the diagnosis and management of CS remain challenging and often require expertise across a range of specialties.1,6,7
Depending on the clinical phenotype of CS, a wide array of interventions, from coronary revascularization, percutaneous or surgical correction of structural heart disease through to mechanical circulatory support (MCS) adjunctive to critical care management, may be required to maintain life-sustaining physiology. Delays in recognition of CS and access to disease-modifying or hemodynamically stabilizing interventions likely contribute to the significant lethality of CS. This may be compounded by heterogeneity of practice within and between hospitals, reflecting a limited evidence base, as well as logistical factors that can delay patient transfer to an experienced center for definitive intervention or hemodynamic assessment and support. These human factor elements are well recognized in other fields of acute care.8 The ideal service would deliver evidence-based or best-practice care efficiently, effectively, and reliably, with equity of access for patients and referrers alike. Care would be multidisciplinary and ideally longitudinal, from resuscitation through to rehabilitation.
As a move towards this model, and in parallel with other time-critical clinical scenarios that have seen quantitative and qualitative improvements in care and process, regional networks of CS care are emerging.9–22 Referrers from regional hospitals are supported by a central shock hub staffed with a designated multiprofessional shock team and acute cardiac care specialists, with all the requisite technologies for diagnostics and patient management.12,14,15 The reliability of escalation to definitive care is ensured by locally developed protocols, with integration of consensus-guideline best practices to ensure timely and appropriate escalation and de-escalation of care, specifically relating to the use of MCS technologies, and mitigation of complications. In an era of increasing incidence of CS with mortality rates that remain between 30% and 40% in contemporary randomized trials and cohort studies, it is hoped that these systems of care will impact primarily on patient-centered outcomes, as well as on the quality and reliability of care and potentially cost-effectiveness.5,23–26
This narrative review outlines improvements from a networked approach to care, discusses a team-based and protocolized approach to CS, reviews the available evidence and discusses the potential benefits, challenges, and opportunities of such systems of care.
Learning from Other Systems
CS patients form a heterogeneous cohort at the thin end of the wedge of those presenting with heart failure or MI. In other groups of critically ill patients, network-based care has led to improved outcomes.9–11 Coordination of ST-elevation MI (STEMI) care is perhaps the exemplar of network-based care. Emergency medical and ambulance services, community hospitals, and tertiary referral centers seamlessly interact to form standardized ‘hub-and-spoke’ STEMI networks to ensure timely reperfusion.9 These networks are supported in the US and UK by societal programs, such as the American Heart Association (AHA) and British Cardiac Intervention Society, to provide quality assurance metrics and mechanisms for quality improvement.
In major trauma, the Advanced Trauma Life Support (ATLS) paradigm emphasizes the need for all centers to be able to provide life-saving initial resuscitation and diagnostics, but also the requirement for the sickest patients to be triaged to tertiary ‘hub’ centers for definitive care. Similarly, the ATLS program incorporates research and injury prevention education. Transitioning to this model in major trauma has resulted in both qualitative service improvements and increased survival.10 Although these systems can result in improved patient outcomes, a key issue in translating these improvements to CS patients is their heterogeneity.9–11 Although STEMI and major trauma are relatively easy to classify and triage, the lack of clear consensus as to what constitutes CS and the difficulty in identifying the CS patient prior to manifestations of multiorgan failure provide particular challenges to introducing streamlined systems of care.
Cardiovascular procedural volumes have been consistently and positively associated with improved clinical outcomes, including survival.28–32 International practice guidelines recommend minimum procedural volumes for hospitals and operators for the maintenance of both accreditation and competency.33,34 In addition to procedural volumes, increased hospital volumes of CS patients are positively associated with improved outcomes in CS.35–38 An analysis of a US payer healthcare database including more than 500,000 CS patients demonstrated that hospitals treating <27 cases of CS each year had an absolute in-hospital mortality increase of 5%, compared with hospitals treating ≥107 cases/year.35 A similar effect was seen in a large sample of AMI CS.36 Mortality in centers with durable left ventricular assist device (LVAD) capability, a likely surrogate of existing experience and expertise in the treatment of CS, was lower than in non-LVAD centers, even after accounting for revascularization rates and the use of mechanical support devices.37 Similarly, data from the greater Paris area showed a relative risk reduction in intensive care unit (ICU) mortality of 24% in CS patients managed in a university hospital compared with a non-university facility.38
Although these volume–outcome relationships will be influenced by confounders, they may also reflect increased access to therapies that may improve outcomes in CS. In the US payer healthcare dataset, CS patients in high-volume centers received higher rates of early revascularization in AMI CS and more frequent escalation to MCS.35 In CS patients undergoing percutaneous coronary intervention, intra-aortic balloon pump (IABP) or MCS use was more common in larger hospitals (>600 beds) and university and teaching hospitals than in community hospitals.39 Similarly, the use of invasive hemodynamic monitoring, which may aid in the identification, management, and prognostication of CS, is almost three time more frequent in urban teaching hospitals than in smaller hospitals.35,40,41
Despite this, 90% of AMI CS patients in a US registry were managed in community hospitals, with 43% treated in low-volume coronary intervention and cardiac surgery centers.24 Another likely contributor to the volume–outcome effect in CS is a ‘failure to rescue’ the sickest CS patients who present to resource-limited, lower-volume centers. Systems of care that foster collaboration between larger and smaller academic and community centers to facilitate the early identification, stabilization, and escalation of CS patients with expedited transfer for definitive management remain an unmet need.
Team-based Care in Cardiogenic Shock
The concept of team-based cardiovascular care is not novel, and heart teams currently support decision making in a range of elective cardiac interventions.34,42,43 A critical element of the management of CS is efficient and reliable alerting of a range of specialists to allow collaborative and streamlined decision making in highly complex and potentially high-resource-utilizing patients, particularly where there is a limited evidence base to drive practice.13–17 A core team of specialists, the shock team, is essential for time-critical decision making, supported by a wider team to ensure holistic follow-on care. The optimal composition of the shock team will depend on local resources, but contemporary shock teams typically comprise an interventional cardiologist, a cardiac critical care physician, cardiac critical care nurses, an advanced heart failure cardiologist, and a cardiac surgeon. Experience and knowledge in the management of CS is vital given the acuity and heterogeneity of presentation.
Although any given shock team may have leadership with overall responsibility for the patient, decisions should be made by consensus with due regard for the expertise and experience of the team members. Ideally the shock team should be capable of providing a response 24 hours a day, 365 days a year, regardless of patient location: emergency department, ICU, catheter laboratory, operating theatre, or off-site hospital. This reliability and resilience can be supported by technology solutions that allow real-time alerts and conference calling with first responders.44 The role of the shock team is to facilitate specific interventions tailored to the etiology of CS and real-time patient physiology, provide expertise that supports locally approved escalation protocols for MCS, and efficiently triage patients to an appropriate care location. Although the evidence base for MCS in cardiogenic shock is incomplete, both European Society of Cardiology and American Heart Association guidelines recommend that patients with CS refractory to first-line therapies undergo evaluation for temporary MCS by a clinical team with experience in MCS technologies.45–47 Therefore, the shock team should have the capability to facilitate emergency institution of MCS where deemed appropriate. This underpins the need for the team to consist of clinicians from interventional cardiology, advanced heart failure cardiology, and cardiac surgery.
Finally, it must be remembered that CS is a condition with an exceptionally high mortality. Even when treatment is optimal and best practices are followed, patients may not survive and survivors may suffer complications and unacceptable functional limitation. The time critical nature of CS requires rapid decision making, often with incomplete premorbid information and an incapacitated patient. As such, it is incumbent on the shock team to ensure that, even when decisions are time critical, the ethical considerations of aggressive, often highly interventional treatments are borne in mind. Routine involvement of palliative care specialists in decision making has been advocated.47 The inclusion of critical care nursing staff and early recourse to palliative care specialists where futility is likely or evident would seem advisable, and consideration of patient or family wishes should be at the forefront of all decision-making.
Protocolized Care in Cardiogenic Shock
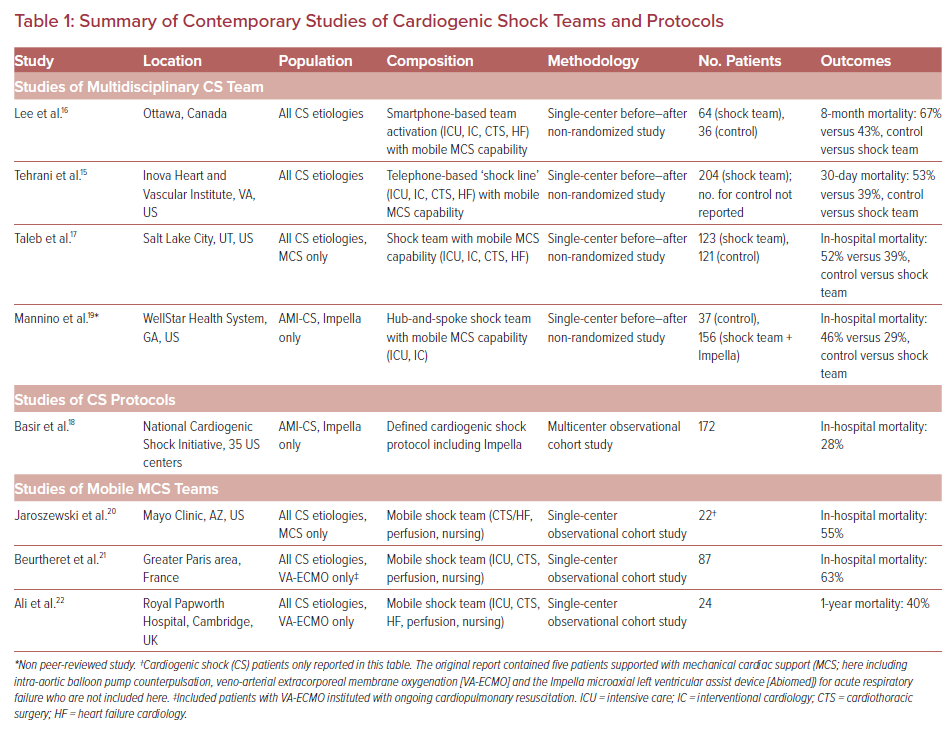
A number of healthcare systems in North America have described the development of shock teams and associated escalation protocols to streamline care and improve outcomes in patients with CS (Table 1). To date, there are no randomized studies on the use of CS teams, and comparisons are limited to historical controls from before–after studies.
The three largest reported studies to date included a total of 391 CS patients treated by multidisciplinary shock teams.15–17 The introduction of a telephone-activated multidisciplinary shock team at the Inova Heart and Vascular Institute in Virginia, accompanied by an institutional protocol for CS management, was associated with a reduction in 30-day mortality from 53% to 23%.14,15 A longer-term survival benefit was also reported from the introduction of a smartphone-activated shock team at the University of Ottawa, which was associated with a reduction in 8-month mortality from 67% to 43%.16 Both these studies introduced multidisciplinary shock teams comprising at least clinicians from critical care, heart failure cardiology, interventional cardiology, and cardiac surgery.
Although the majority of clinical decision making may be conducted by the core team, the involvement of the wider patient care team cannot be underestimated. Care is improved when bedside nursing staff and allied health professionals (including physiotherapy, occupational therapy, pharmacy, nutrition and dietetics, and perfusion staff) have experience of treating CS patients, including those on MCS. Because CS patients often develop multiorgan dysfunction and a systemic inflammatory response syndrome, other available specialist physicians with an interest and experience in CS, including, but not limited to, nephrology, hematology, electrophysiology, anesthesia, and stroke medicine, also form part of the wider circle of individuals involved in the care of CS patients.
Other studies of shock teams have similarly reported the outcomes of CS patients treated with MCS as part of a multidisciplinary team approach. A study from the University of Utah compared 123 patients treated with short-term MCS by a shock team to a historical cohort of consecutive MCS patients without a team-based approach, reporting a reduction in in-hospital mortality from 52% to 39%.17 The National Cardiogenic Shock Initiative included 172 patients with AMI CS managed at 35 US centers with a protocol emphasizing early revascularization supported by pre-percutaneous coronary intervention (PCI) MCS, with a percutaneous microaxial flow left ventricular device (Impella, Abiomed) guided by hemodynamic data.18 This approach improved survival from 50% in a historical cohort to 72%, although the use of a shock team to support decision-making was not specified.16 A single-center study from the WellStar Health System in Georgia reported before and after hospital mortality of 46% and 29%, respectively, in a non-peer-reviewed study of a hub-and-spoke CS network in 156 patients treated with Impella.19
Although caution must be exercised in drawing conclusions from observational before–after studies, there is both a compelling rationale for the role of shock teams and a growing body of data to support patient-level benefit. Which specific aspects of shock protocols are likely to be essential and affect mortality remains unclear. Protocols may standardize clinical approach within and across institutions. However, they may fail to address complex issues in real-world settings or allow individualization of patient care. Despite recent advances in classification, CS remains ill-defined and incorporates a range of phenotypes that cannot be encapsulated by limited physiological and biochemical parameters alone.6,48,49 The role of early, pre-revascularization MCS in AMI CS remains unproven in randomized trials, as does the role of right heart catheter data to guide decision making and escalation to MCS, despite supportive observational data.45,50,51 Given the burden of complications of MCS and their effects on mortality, protocols pertaining to large-bore vascular access, anticoagulation, and MCS weaning and removal would seem prudent. Therefore, the role of shock teams may be to synthesize and analyze a locally (or regionally) defined minimum dataset of clinical, biochemical, imaging, and physiological parameters to guide the optimal timing of escalation to MCS, as well as device and patient selection.
Shock Hubs Within Networks of Cardiogenic Shock Care: Towards a 21st Century System of Care
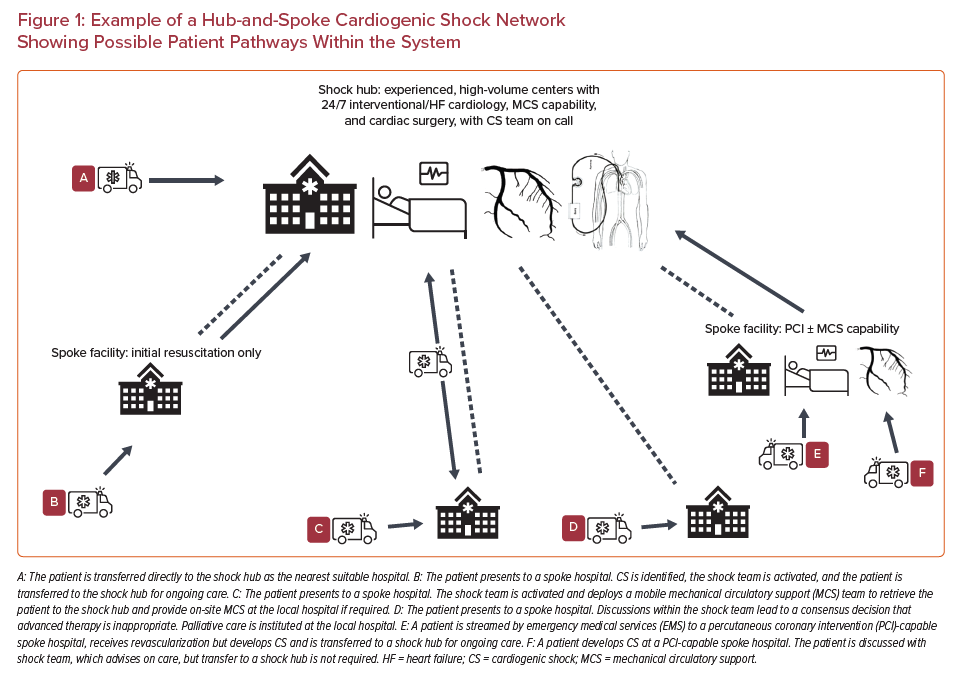
The development and maturation of shock teams within high-volume cardiac centers is an intuitive first step. However, the extension of this expertise throughout a more formative network of CS care is required to deliver care equitably for both patients and referrers, and to improve patient-level outcomes at scale. This approach to CS care level has been advocated by societal guidelines and consensus statements.47,50,52 Proposed definitions of the requisite components of a network are outlined in Table 2. Given the prevalence of AMI CS within the cohort of CS patients as a whole, existing pathways to prioritize primary PCI in STEMI are a natural starting point, while also recognizing that non-AMI-related CS patients will have discrete needs, and may present through different pathways throughout the network. The outline of a potential regional shock network is shown in Figure 1, with key clinical logistical and organizational elements outlined in Table 3. Peripheral-spoke hospitals may provide a range of services, including primary PCI and/or percutaneous MCS therapies, but many will only have the facility to provide initial assessment and resuscitation of patients. Because most CS patients will not present to a shock hub, the approach should be collaborative, with representatives from all types of facilities involved in all aspects of networked care delivery from inception.24,35
For the sickest CS patients (Society for Cardiovascular Angiography and Interventions stage D or E6), survival may depend on rapid access to MCS to sustain physiology; when such patients present at spoke centers without the ability to institute MCS locally, they will often also be too unstable to safely transfer to definitive care. A number of single-center studies have described mobile MCS teams set up to retrieve patients from off-site hospitals after institution of MCS.20–22 These cohorts, although small, report higher mortality than studies of systems where patients are moved to an MCS center and escalated according to local protocols. Whether this reflects a selection bias, the benefit of care in an experienced center, or the role of local protocols in patient selection is unclear. Nonetheless, based on these data, mobile MCS is safe and feasible.20–22 The need for transport infrastructure within a CS network will depend on local geography and healthcare organization. The key standard is that the patients throughout a network have equitable and timely access to specialist support and associated interventions such as MCS.
Given the overlap between CS and cardiac arrest, and the emerging evidence base for extracorporeal membrane oxygenation in the treatment of refractory sudden cardiac arrest, extracorporeal cardiopulmonary resuscitation (eCPR) capability is likely to become a prerequisite for shock hubs.53,54 Although much of the infrastructure required for CS systems overlaps with that required for eCPR, eCPR demands 24/7 on-site expertise for patient selection and cannulation that may require the triage of patients to select geographical hubs or even mobilization of cannulation teams to the patient in the field. The resource implications of eCPR service provision are significant and should be balanced with the provision of optimal CS care at scale within shock hubs.
As CS systems of care evolve, it is vital to ensure continued stakeholder engagement, integration, and quality improvement inclusive across the entire patient journey. Clear pathways and protocols should be developed for referral, acceptance, transfer, and repatriation. Reimbursement will vary across healthcare systems, but an analysis of cost and the cost-effectiveness of the shock team and a networked approach have not been adequately described. Despite the challenges of clinical governance across different institutions, the limited evidence base to guide interventions and high complication rates with MCS should mandate an open culture of learning across the network to ensure that ‘failure to rescue’ scenarios are identified and mitigated and that protocols and best practices are iterated as local experience and published data evolve, complications are minimized, and outcomes are optimized.
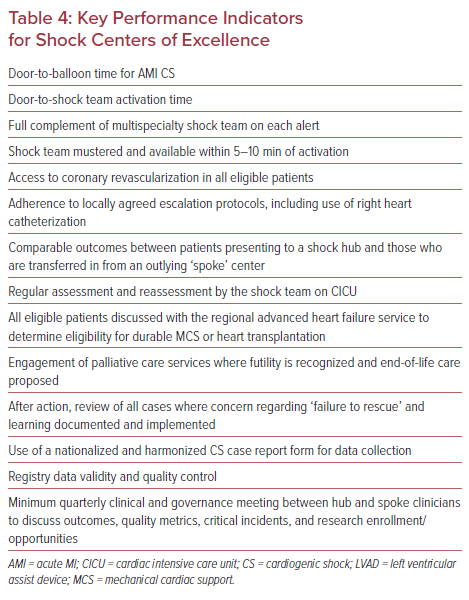
These quality improvement efforts require routine and robust data collection. Networked data collection and integration would also facilitate longer-term follow-up data analysis, including post-discharge healthcare utilization and quality of life metrics, and a shift towards a holistic model of CS care from resuscitation to recovery. Nonetheless, although local registries provide valuable information about participating network sites, simple comparisons between hospitals are not sufficiently reliable for reporting trends in demographics, access to interventions, and patient outcomes. Ultimately, data collection and analysis at scale could be achieved via a harmonized national case report form. This model has been adopted by the American Heart Association through the Get With the Guidelines initiative, which has led to transformation across several facets of acute cardiac care and could be a template for development of a nationalized CS registry given the overlap with existing programs in AMI and out-of-hospital cardiac arrest.55–57 This model may assist in validated designation of CS ‘centers of excellence’ using putative key performance metrics (Table 4).
Beyond quality improvement, research infrastructure and efficient approaches to informed consent should be a defining criterion of shock hubs. Given the historical challenges of patient recruitment to CS trials, it is imperative that with centralization of the sickest CS patients to shock hubs, the opportunity for clinical trial enrolment is leveraged.58 Research activity is associated with better clinical outcomes in acute care and should form part of shock network activity.59
Indeed, it should be remembered that with the exception of early revascularization in AMI CS, none of the interventions likely to be instituted by CS teams, systems, or shock hubs (e.g. MCS, invasive hemodynamic monitoring, or protocolized care) have been shown to improve outcomes in randomized trials.60 Given the clear need to improve the evidence base for CS, the authors suggest that a key role of the shock team should be identification of eligible patients for future observational and randomized, intervention-based trials.
Conclusion
Mortality in cardiogenic shock remains unacceptably high. As has been demonstrated in other diseases, a multidisciplinary protocolized team-based approach with regional hub-and-spoke networks of care built around high-volume, experienced centers is likely to improve outcomes. The existing data, although incomplete, support this approach. The development of processes to ensure equitable access to CS care across geographical and institutional boundaries requires a consensus approach with a robust educational and governance structure alongside clinical pathways. As the clinical care of CS patients continues to improve, the creation of CS networks provides a unique opportunity to replicate the success of other acute cardiovascular care pathways and realize improved outcomes for this disease, as well as engagement in future clinical trials.