Moderate-to-severe tricuspid regurgitation (TR) is estimated to affect more than 1.6 million people in the US. However, fewer than 8,000 tricuspid valve operations are performed each year.1,2 There is clearly an unmet need for a tricuspid valve (TV) intervention. There has been rapid development in the field of transcatheter aortic and mitral valve interventions and percutaneous management of tricuspid valve disease has also evolved substantially in recent years. With increasing evidence of a poor prognosis associated with untreated severe TR, the once ‘forgotten valve’ has gained more attention.3 In this article, we provide a review of tricuspid regurgitation and transcatheter edge-to-edge repair.
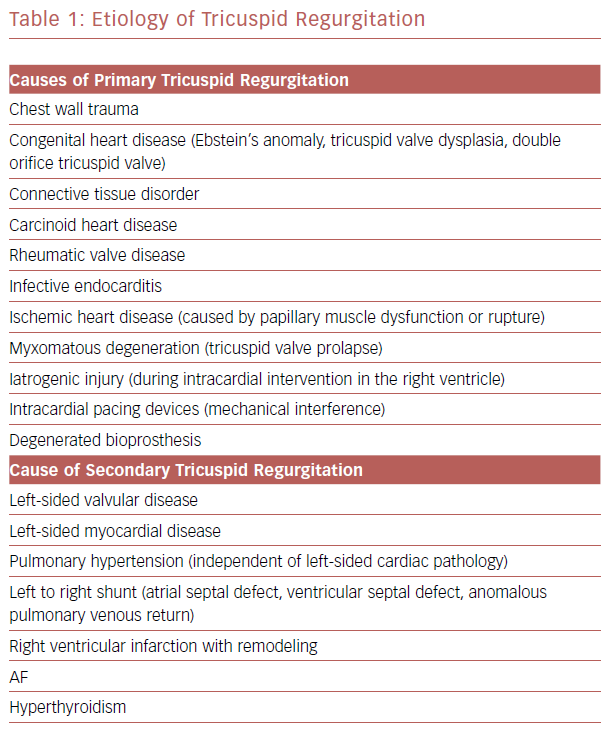
Etiology of Tricuspid Valve Regurgitation
The most frequent etiology of tricuspid valve disease is secondary (or functional). In a study of 242 consecutive patients diagnosed with severe TR by echocardiography, 90% of the patients were defined as secondary TR.4 Secondary TR refers to TR with anatomically normal leaflets and chords with regurgitation occurring due to right atrium and ventricle dilatation, annulus dilatation, and leaflet tethering leading to malcoaptation. The common causes of secondary TR are left-side myocardial or valvular heart disease and pulmonary hypertension. Atrial remodeling associated with atrial fibrillation and annulus dilatation is also a notable cause of significant TR5.
Primary TR accounts for 10% of severe TR, and is caused by the process that directly affect the leaflets of the tricuspid valve. The common causes include congenital abnormality, tricuspid prolapse, trauma, endocarditis, and intragenic injury by myocardial biopsy or intracardiac leads.6,7 Table 1 lists possible causes of TR.
Image Evaluation
The primary method of evaluating tricuspid valve regurgitation is echocardiography. Transthoracic echocardiography (TTE) can allow the etiology and severity to be evaluated by measuring the dimension of the right ventricle, right atrium, and tricuspid annulus. Other important information includes tricuspid valve motion, coaptation, Doppler color flow of the TV, right ventricular function, pulmonary artery pressure, and concomitant left-sided valvular or myocardial disease.
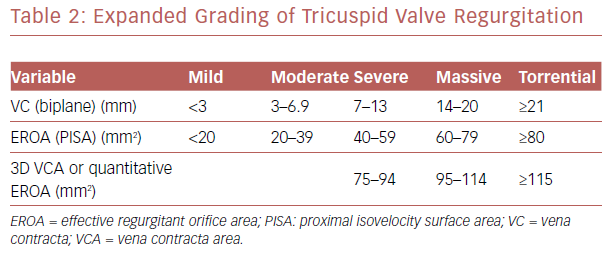
A comprehensive echocardiographic assessment is crucial to the grading of tricuspid regurgitation severity and will inform the treatment strategy. The European Society of Echocardiography and American Society of Echocardiography have suggested that grading of severity as mild, moderate, or severe should be made by using the combination of structure, qualitative Doppler, semiquantitative, and quantitative measures.8,9 However, in a recent transcatheter trial, patients were enrolled with late stage TR and most of them developed severe right-side heart failure.
The current grading system often fails to take into account the ‘torrent’ nature of TR severity in people with advanced diseased. Hahn et al. proposed increasing the grades to include very severe (or massive) and torrential (Table 2) to better characterize the severity of TR.10 Echocardiography measurement also provides information of outcome predictors. Significant annulus dilatation defined as diastolic diameter ≥ 40mm is a predictor of severe late TR after mitral valve surgery.11,12 Transesophageal echocardiography (TEE) is considered when there is an inadequate image found using TTE. The most important role of TEE is to guide steering of the transcatheter device during transcatheter tricuspid valve intervention.
CT imaging has an important role in pre-procedural planning for tricuspid valve intervention. Detailed information of the end-diastolic leaflet tenting distance which shows correlation to TR severity and outcome after surgical repair, tricuspid annulus dimension, right ventricular volumes, and function can all be ascertained using CT.13 Cardiac MRI provides excellent spatial resolution of right heart structures and it is the gold standard method for quantifying right ventricular volumes and function.14
Prognosis
TR is independently associated with increased cardiac events and mortality in patients with congestive heart failure.15 In a retrospective study of 5,223 patients, after controlling left ventricular ejection fraction, right ventricular dilation and dysfunction, and pulmonary artery systolic pressure, mortality increased with increasing severity of TR.16 The 1-year survival rate was 91.7% with no TR, 90.3% with mild TR, 78.9% with moderate TR, and 63.9% with severe TR. In patients with left ventricular dysfunction, those who developed severe TR were in a high-risk subset.17 Another study involved 353 patients with isolated TR and excess mortality and cardiac event rates were found to be associated with severe regurgitation.18 These studies indicate that TR has been independently associated with a worse prognosis in patients with or without left ventricular systolic dysfunction and with or without clinical heart failure.
Surgical Treatment
Current guidelines recommend that tricuspid valve surgery is indicated for patients with symptoms of severe primary TR that are not responsive to medical treatment.19 Surgery is also considered in asymptomatic or mildly symptomatic patients with severe primary TR and progressive right ventricular dilatation or dysfunction. However, although the utilization has increased over time, isolated TV surgery remains rare. In a large US national registry, a total of 5,005 isolated TV operations were performed over 10 years with an in-hospital mortality rate of 8.8%.20
Tricuspid valve surgery is indicated in patients with primary or secondary severe TR undergoing left-sided valve surgery, surgery is also considered in patients with moderate primary TR who are undergoing left-side valve surgery. There is an increasing amount of evidence to support a more aggressive surgical approach for secondary TR. In addition, because of the hazards associated with reoperation, concomitant tricuspid valve repair is reasonable for patients with non-severe TR.21
Current guidelines suggest surgery should be considered in patients with mild to moderate secondary TR undergoing left-side valve surgery with a dilated annulus ≥40mm, and surgery may be considered in the absence of annulus dilatation when previous right-heart failure has been documented.19 After previous left-sided surgery, reoperation should be considered for persistent symptoms or progressive right ventricular dilatation caused by secondary severe TR.
Reoperation of the tricuspid valve is associated with poor outcomes and an early mortality rate of 10–25%, probably because this group of patients are usually already in late-stage TR with severe right ventricular dilatation and organ damage.3 Tricuspid annuloplasty with suture or ring is the main technique for secondary TR because annular dilatation is the most common mechanism. Ring annuloplasty is the preferred technique due to increased durability compared with suture technique.22
Transcatheter Tricuspid Valve Intervention
Despite guidelines that suggest surgical treatment, many patients with severe TR are still treated conservatively due to the high risk of surgery. Patients with severe TR and previous open-heart surgery are often deemed at prohibitive operative risk for reoperation. Patients in late stage of secondary TR and progressive right heart dysfunction despite medical treatment are also usually considered as high risk for isolated TR repaired. In recent years, a novel transcatheter valvular treatment option for treating TR has been developed. Patients with prohibitive operative risk may benefit from the transcatheter treatment as an alternative.
Transcatheter Edge-to-edge Repair
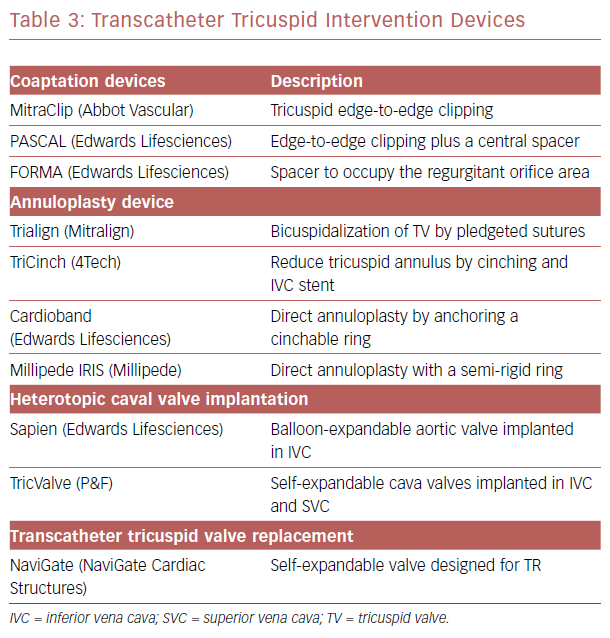
Multiple technologies for transcatheter tricuspid intervention are currently under preclinical or initial clinical evaluation. Those treatments can be grouped into coaptation, annuloplasty device and valve implantation device (Table 3). Taramasso et al. reported initial results of The TriValve registry suggesting the feasibility of transcatheter tricuspid valve therapy.23 Among those novel transcatheter tricuspid intervention techniques, the edge-to-edge repair with off-label use of MitraClip system (Abbott Vascular) has become the most common choice because of the system’s wide availability and operator familiarity.
Surgical Perspective on Transcatheter Edge-to-edge Repair
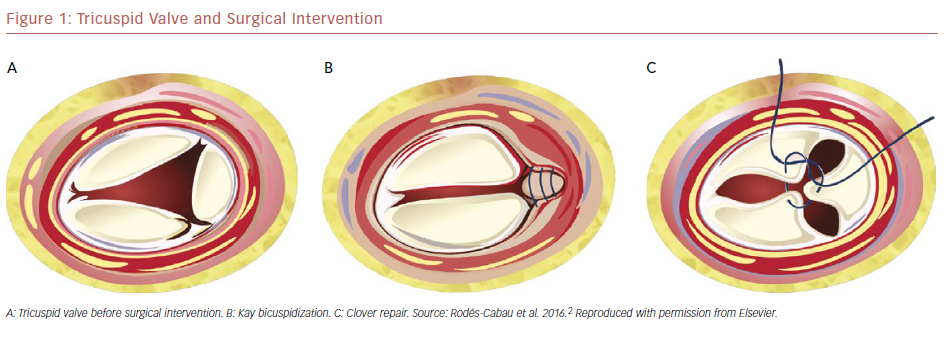
Surgical edge-to-edge tricuspid repair was reported early by Maisano et al. to treat traumatic tricuspid regurgitation.24 Currently, surgical tricuspid annuloplasty is the foundation of surgical treatment of secondary TR. Two principal methods of surgical annuloplasty are suture and ring techniques. Kay bicuspidization by surgical plication of the posterior leaflet is one cornerstone of suture annuloplasty (Figure 1b).25 The clover technique involves suturing the three leaflets edge to edge and has been used to treat severe TR due to severe prolapse or tethering (Figure 1c).26
Transcatheter edge-to-edge repair technique can produce similar result as a modified Kay technique or the clover technique. Based on the concept of surgical correcting annular dilatation to improve leaflet coaptation, percutaneous edge-to-edge tricuspid clipping is a sound technique.27 A combination of transcatheter annuloplasty device and edge-to-edge repair is also a possible therapeutic option.
Preliminary Experience of Transcatheter Edge-to-edge Repair
The first case reports of transcatheter edge-to-edge repair published in 2016 demonstrated its technical feasibility and procedural safety for patients with severe primary or secondary TR with a prohibitive operative risk.28,29 Hammerstingl et al. reported three consecutive patients treated successfully for significant TR in advanced stages of right heart failure and multiorgan damage.30 Reduction of measured effective regurgitation orifice area (EROA) was observed in all the patients. Other clinical benefits included an increase in left ventricular stroke volumes and relief of clinical symptoms.
Image and Technique During Procedure
Although preliminary experience has shown the feasibility of tricuspid valve clipping, intraprocedural guidance remains a major issue. Procedural guidance with 3D TEE is a fundamental tool to guide the procedure. However, the three-leaflet configuration of TV and often suboptimal TEE quality contributes to the increased technical complexity of TV clipping. A combination of TEE, TTE and intracardiac ultrasound together with fluoroscopy are used to achieve better visualization. Communication between the echocardiographer and interventionalist is fundamental during the procedure and the nomenclature in the tricuspid valve position while steering the device is important. Taramasso et al. proposed to use aortic-posterior and septal-anterior directions in 3D TEE surgical view to better describe the orientation of TV during steering.31
The use of the MitraClip system for the TV raises some issues because the device was designed specifically for the mitral valve. One technical issue is the proximity of the inferior vena cava orifice to the tricuspid valve coaptation line and atrial septum, which makes steering of the MitraClip delivery system perpendicular to the tricuspid valve plane more difficult. Daniel et al. proposed to insert the clip delivery system 90° counterclockwise from its standard locking position into the sheath to accommodate the system in the tricuspid valve plane with an orthogonal orientation.32 In the future, the technique must be refined, and the device be modified for dedicated use for tricuspid valve disease.33
The optimal clip position strategy for reducing the EROA by closing the valve remains to be established. In an ex vivo pulsatile heart model study, grasping the septal and anterior leaflets allowed for the best post-procedural outcome.34 Two-clip implantation involving the septal-anterior and septal-posterior position also increased cardiac output. Tricuspid bicuspidization using a modified zipping technique has been proposed.32 The first clip is placed into the anteroseptal commissure, which is the easiest to target and the subsequent clips are placed in wards close proximally to the previous clips.
A retrospective study of 69 consecutive symptomatic patients with secondary TR treated by MitraClip compared two-clip placement strategies, the triple-orifice technique (TOT) and bicuspidization technique.35 Two clips are placed centrally between the septal and anterior tricuspid leaflet, as well as the septal and posterior tricuspid leaflet in TOT. The procedural success rate was 93% in the TOT group and 84% in the bicuspidization group. A persistent reduction of TR ≤2 was demonstrated in both groups and the septolateral tricuspid annulus diameter had a greater reduction in the TOT group.
Patient Selection
Based on current studies, most patients who undergo transcatheter edge-to-edge repair were deemed high risk for surgery. In many cases the procedure is performed as a compassionate treatment (the use of a new, unapproved drug to treat a seriously ill patient when no other treatments are available), so it is difficult to assess which patient is the perfect target for transcatheter edge-to-edge repair. Patients with poor echocardiography visualization of the tricuspid valve, with concurrent tricuspid valve stenosis and unsuitable anatomy were excluded from most studies, but there is no established consensus on unsuitable anatomy. A recent study proposed the conditions that make leaflet grasping unlikely: an EROA >1.5 cm2, a TV coaptation defect >15 mm, and TR caused by markedly restricted leaflet mobility due to pacemaker or implantable cardioverter defibrillator leads across the TV.36 Advanced age, severe impaired left or right ventricular dysfunction, pulmonary hypertension, and concomitant mitral regurgitation are not absolute contraindications in current practice. Multidisciplinary discussion by the heart team is mandatory for each case selection.
Current Outcomes from Clinical Experience
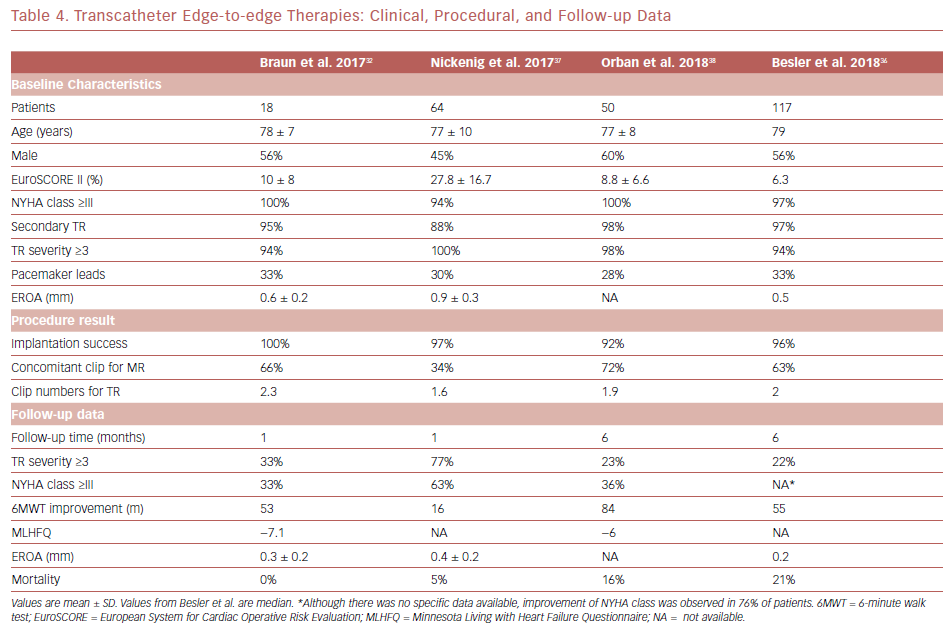
More than 650 procedures using the MitraClip system in tricuspid valve treatment have been performed worldwide. Increasing evidence has been published that assesses the outcome and safety of the procedure. In a multicenter European registry, 64 high-risk patients with severe TR were treated with the MitraClip system.37 The MitraClip device was successfully implanted in the tricuspid valve in 97% of the cases. TR was reduced by at least one grade in 91% of the patients after the procedure. Significant reduction in EROA, vena contracta width and regurgitation volume were observed. New York Heart Association (NYHA) class and 6-minute walk test distance were improved significantly at 30 days post-procedure.
A case series with 18 consecutive patients with symptomatic moderate-to-severe TR treated using the MitraClip system was reported.32 Implantation success was achieved in all patients and no major adverse cardiac events occurred in hospital. There was a significant reduction in the percentage of patients with TR ≥3 from 94% to 33% after the procedure and 89% of patients had an improved NYHA functional class at 30 days.
Orban et al. reported 6-month follow-up result of 50 patients with right-sided heart failure and severe TR treated with the MitraClip system.38 Among them, 36 patients received combined mitral valve and tricuspid valve clipping. A persistent reduction of at least one TR grade was achieved in 90% of patients after 6 months. NYHA class improved in 79% of patients, and 6-minute walk distance increased by 44%. The improvement was observed in patients treated with isolated tricuspid valve and concomitant mitral valve plus tricuspid valve repair.
Recently, Besler et al. reported median 6-month follow-up of 117 patients who underwent transcatheter edge-to-edge repair with 96% success rate for clip implantation.36 After the procedure, 81% of patients had TR reduction of more than one grade, mean vena contracta reduced from 9 mm to 5 mm (p<0.01) and median TR EROA from 0.5 mm2 to 0.2 mm2 (p<0.01). Improvement of NYHA functional class was shown in 76% of patients and 6-minute walk test distance improved by 29%. In the study, a successful procedure (TR reduction ≥1) was an independent predictor of reduced mortality and heart failure hospitalization. Five echocardiographic parameters were developed to predict procedural success: small TR EROA, a smaller TR tenting area, TR vena contracta, a smaller TV coaptation gap and a central and anteroseptal TR jet location. A coaptation gap of 7.2 mm has been proposed as a cut-off value to assist in deciding whether a patient is anatomically suited for transcatheter clipping.
The clinical and procedural features of patients treated with edge-to-edge repair mentioned above are outlined in Table 4.
MitraClip XTR
Recently, the new generation MitraClip XTR device has been used for tricuspid valve repair. The new device has clip arms and grippers that are 3 mm longer than the MitraClip NTR clip, facilitating easier grasping of the leaflets, making it a preferred choice to treat severe TR with a large coaptation gap. A dedicated tricuspid clip delivery system has been developed and is under evaluation. The evaluation of treatment with an Abbott transcatheter clip repair system in patients with moderate or greater tricuspid regurgitation (TRILUMINATE) trial is a prospective, multicenter, single-arm study that has enrolled 85 patients with symptomatic TR treated with a novel Abbott transcatheter tricuspid valve repair system. Early results from the first 30 patients have been reported recently with a 100% device implantation success rate.39 The early results show effectiveness for TR reduction and improvement of symptoms and quality of life after 30 days follow-up. A low rate of major adverse event without mortality through 30 days has been reported.
PASCAL
The other transcatheter edge-to-edge repair device is PASCAL (Edwards Lifesciences), which had been developed to treat mitral regurgitation. The device combines a 10 mm central spacer with two paddles and clasps that attach the device to the valve leaflets. The device is repositionable and recapturable.
The first-in-human study enrolled 23 patients with moderate-to-severe mitral regurgitation using the PASCAL system showed the feasibility with a high rate of technical success and reduction of mitral regurgitation severity.40 The first 12 cases of using PASCAL to treat severe TR were reported recently.41,42 Among 12 patients, the mean tricuspid septal-lateral annulus diameter is 52.3 mm and mean maximum coaptation gap size is 7.3 mm. After the procedure, 92% of patients had TR reduction of more than one grade. Clinical improvement in NYHA class and reduced mean values of TR grade have also been demonstrated. This device offers a new option for patients who have severe TR with large coaptation gaps and leaflet tethering. Further research to evaluate efficacy, safety and durability is needed.
Conclusion
Although the growing evidence addresses the clinical importance of significant tricuspid valve regurgitation, there are still an extremely large number of untreated patients with moderate-to-severe TR. Transcatheter tricuspid intervention is an appealing option for the treatment of this group of patients. Transcatheter edge-to-edge repair devices are currently the most popular among these novel technologies for percutaneous treatment of TR.
Transcatheter edge-to-edge repair is still at a preliminary stage. Imaging during intervention is a challenge and procedural success is dependent on the experience of the interventionalist and echocardiographer and close interaction between them. Initial results from the procedure have showed feasibility and safety. Increasing data have shown the benefits of the procedure on clinical outcomes. Most studies reported significant improvement in TR grade, symptoms, and quality of life. A recent retrospective study demonstrated lower mortality and hospitalization after a successful procedure.36 We still need more evidence to understand optimal patient selection, correct timing of intervention, durability of the device, and long-term outcome of the treatment.